

Lysosomal enzyme
Discovery of the lysosome
In 1949, de Duve, a scientist at the Institute of Cytopathology in Brussels, Belgium, homogenized rat liver tissue and isolated various organelles with a view to identifying those related to enzymes of glucose metabolism, and hypothesized that a new organelle existed in the cell based on the results of the experiments. de Duve, in collaboration with Novikof, observed this organelle for the first time with an electron microscope in 1955, and it was named lysosome in 1956. He shared the 1974 Nobel Prize in Physiology or Medicine with his colleagues, electron microscopists Claude and Paradis.

Structure of lysosome
So far found only in animal cells, the lysosome is a vesicle containing a series of acidic hydrolyzing enzymes surrounded by a single lipoprotein membrane in the cytoplasm. It is a vesicle-like structure surrounded by a single membrane and is a dynamic structure. In different types of cells, lysosomes vary in shape and size. Generally, they are round vesicles with a diameter of 0.25~0.8μm, containing a variety of acidic hydrolyzing enzymes, which can decompose a variety of exogenous or endogenous macromolecules. Therefore, lysosomes have been compared to the "enzyme warehouse" and "digestive system" in the cell.
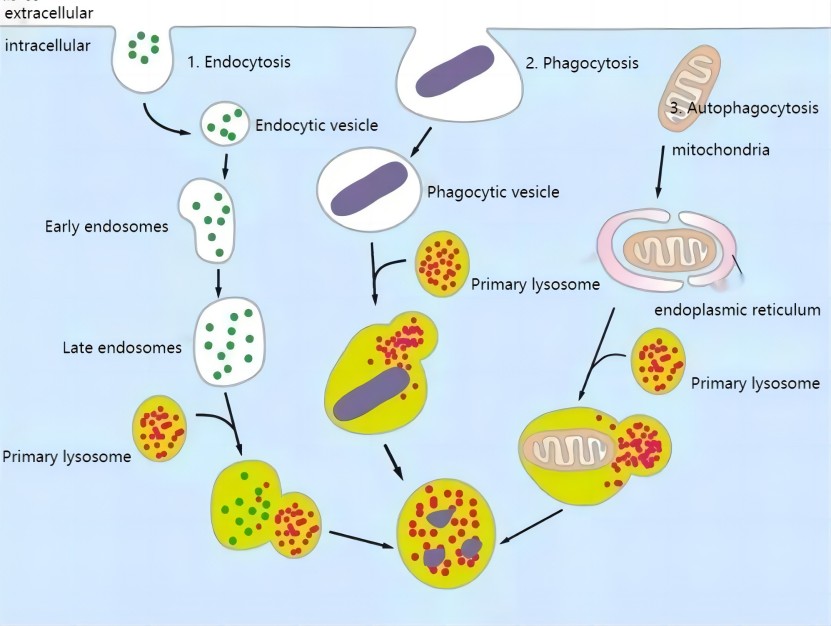
Classification and formation of lysosome
The formation of lysosomes is a rather complex process. It is generally believed that the enzyme in the lysosome is synthesized by the ribosomes on the rough endoplasmic reticulum and transported to the Golgi apparatus, where it is processed, sorted and concentrated, coated with an outer membrane, forming vesicles, and then leaving the Golgi complex, which contains only the hydrolase but not the catalyzed substrate and is called the primary lysosome. Primary lysosomes fuse with intracellular bodies, phagosomes, and autophagosomes to form complexes, and the acidic hydrolases in the lysosomes play a role in the gradual digestion of intracellular bodies and phagosomes, etc. At this time, the lysosomes contain not only hydrolases, but also a large number of catalyzed substrates, and they are a kind of lysosomes that are undergoing digestion, which is called secondary lysosomes. After the completion of digestion in the secondary lysosome, the enzyme activity becomes weak or even lost, and only undigested residue remains, which is called residual body.
Lysosomal Enzyme Related Indicators Available from ELK Biotechnology
|
|
|
Catalog No. |
Product Name |
Size & Price(48T/96T) |
UniProtKB |
|
ELK3300 |
Human ACP2(Acid Phosphatase 2, Lysosomal) ELISA Kit |
$458/$320 |
P11117 |
|
ELK2328 |
Human ACP3(Acid Phosphatase 3, Prostatic) ELISA Kit |
$458/$320 |
P15308 |
|
ELK9590 |
Human TRACP-5b(Tartrate Resistant Acid Phosphatase 5b) ELISA Kit |
$458/$320 |
P13685 |
|
ELK1930 |
Human RNASE1(Ribonuclease A) ELISA Kit |
$458/$320 |
P07998 |
|
ELK1791 |
Human DNASE1(Deoxyribonuclease I) ELISA Kit |
$458/$320 |
P24855 |
|
ELK1911 |
Human CTSK(Cathepsin K) ELISA Kit |
$458/$320 |
P43235 |
|
ELK4714 |
Human ARSA(Arylsulfatase A) ELISA Kit |
$458/$320 |
P15289 |
|
LK2772 |
Human GUSb(Glucuronidase Beta) ELISA Kit |
$458/$320 |
P08236 |
|
ELK4035 |
Human HAT1(Histone Acetyltransferase 1) ELISA Kit |
$458/$320 |
O14929 |
|
ELK9575 |
AcCoA(Acetyl CoA) ELISA Kit |
$458/$320 |
/ |
|
ELK4042 |
Human HDAC1(Histone Deacetylase 1) ELISA Kit |
$458/$320 |
Q13547 |
|
ELK2950 |
Human HAase(Hyaluronidase) ELISA Kit |
$458/$320 |
/ |
Enzymes in lysosome
Type
More than 60 acid hydrolases have been identified in lysosomes, including enzymes that hydrolyze proteins, sugars, lipids, and other substances. Such as acid phospholipase, histone protease, ribonuclease, hyaluronidase, phosphotransferase, β-galactosidase, and aromatic sulfate lipases A and B. Most of the enzymes in lysosomes are glycoproteins, but there are exceptions, e.g., most of the enzymes in the lysosomes of murine hepatocytes and renal cells are lipoproteins.
Source
The enzymes in lysosomes are similar to the formation of secretory proteins. They are all immature enzymes synthesized by ribosomes attached to the endoplasmic reticulum, and then enter the rough endoplasmic reticulum under the guidance of the signal peptide, and connect some enzymes after preliminary processing. Sugar groups, such as glucose, mannose, and N-acetylglucosamine, etc., the endoplasmic reticulum transports these more mature enzymes to the Golgi apparatus through the formation of vesicles, and the reprocessing of the Golgi apparatus, such as the N-acetylglucosamine residue The base is grafted onto the mannose residue, etc., to form a mature enzyme.
Feature
The membrane proteins of lysosomes are mostly glycoproteins, and the inner surface of the lysosome membrane is negatively charged, so it helps the enzymes in the lysosomes to maintain a free state, which is of great significance for the normal function of the cells and prevent the cells themselves from being digested.
All hydrolytic enzymes are most active at around pH 5.0, but the pH in the surrounding cytoplasm is about 7.2, and the lysosome membrane contains a special transport protein that can use the energy of ATP hydrolysis to pump H+ in the cytoplasm into Lysosomes to maintain a pH of 5; only when the hydrolyzed substances enter the lysosomes, the enzymes in the lysosomes can perform their decomposition. Once the lysosomal membrane is damaged, hydrolytic enzymes escape, which will lead to cell autolysis.
Functions of lysosome
Intracellular digestion
For higher animals, the nutrients of cells mainly come from small molecular substances in the blood, while some macromolecular substances enter cells through endocytosis, such as endocytosis of low-density lipoprotein to obtain cholesterol. For some unicellular eukaryotes, the digestion of lysosomes is even more important. For example, paramecium digests the food or pathogenic bacteria that have been swallowed into the cells, and the available nutrients enter the cytoplasmic matrix for various metabolic activities, and the residue is discharged through exocytosis.
Autolysis (Apoptosis)
Remove degenerated cells and dead cells during development through autolysis to ensure normal cell growth and development. The process of ontogeny often involves the transformation or reconstruction of tissues or organs, such as the metamorphosis of insects and frogs, the degeneration of feet, and the disappearance of tails, etc. This process is realized under the control of genes, which is called programmed cell death ( Apoptosis), these cells destined for elimination bud out to form apoptotic bodies, which are engulfed by macrophages and digested by lysosomes, thereby clearing unwanted cells.
Autophagy
Remove useless macromolecules and aging organelles in cells. The half-life of many biological macromolecules is only a few hours to a few days. For example, the average lifespan of mitochondria in liver cells is about 10 days, which requires lysosomes to engulf and digest them.
Defensive role
All leukocytes contain lysosomal granules, which can destroy invading microorganisms. For example, phagocytes can engulf pathogens and process, kill or degrade them in lysosomes. However, some pathogenic bacteria (for example, Bacillus leprae, Mycobacterium tuberculosis, etc.) are resistant to the action of lysosomal enzymes and thus can survive in macrophages.
Involved in the regulation of secretory processes
Studies have found that when the secretion of prolactin from the rat pituitary gland is inhibited, lysosomes fuse with a part of the secretory granules in the cells, digest and degrade them to eliminate excessive hormones in the cells, this phenomenon is called granulocytes or secretory autophagy. Granulolysis occurs in almost all cells that secrete protein and peptide hormones. In this way, cells can effectively regulate the secretion of hormones, such as processing thyroglobulin into active thyroxine.
Formation of the acrosome of the sperm
The acrosome of sperm is also a kind of lysosome in essence. During the fertilization process, the enzyme in the acrosome is released outside the cell, which can digest the follicle cells and the zona pellucida around the egg, forming a channel for the sperm to enter the egg, and facilitate the sperm to enter the egg cell to achieve the purpose of fertilization.


