

Mechanism of ferroptosis in the treatment of liver fibrosis
In 2012, the team of Brent R. Stockwell at Columbia University first proposed a new type of programmed cell death called ferroptosis. Since then, the upsurge of ferroptosis-related research has been increasing year by year. In 2012, the team of Brent R. Stockwell at Columbia University first proposed a new type of programmed cell death called ferroptosis. Since then, the upsurge of ferroptosis-related research has been increasing year by year.
what is ferroptosis?
Unlike apoptosis and autophagy, ferroptosis is an iron-dependent process. The essence of ferroptosis is the depletion of glutathione (GSH) and the reduction of glutathione peroxidase 4 (GPX4) activity. When glutathione peroxides cannot be catalyzed by GPX4, lipid peroxides cannot be metabolized, thereby destroying the integrity of cell membranes and leading to cell death. Ferroptosis has its own unique features in Morphology, Biochemistry and Genetics.
Morphological characteristics
It's characterized by reduced mitochondrial volume, increased bilayer membrane density, decreased or disappeared mitochondrial cristae, and ruptured mitochondrial outer membrane; normal nuclear size without chromatin aggregation.
Biochemical characteristics
Iron accumulation, lipid peroxidation, activation of mitogen-activated proteases (MAPKs) system, inhibition of cystine-glutamate antiporter (System Xc-, a heterodimer composed of SLC3A2 of SLC7A11) decreased cystine uptake, depletion of glutathione (GSH), increased oxidation of NAPDH, release of arachidonic acid mediators (such as 11-HETE and 15-HETE), etc.
Immune characteristics
Releases damage-associated molecular patterns (DAMPs) , promote inflammatory responses.
Iron metabolism and ferroptosis
Iron is an indispensable trace element in the human body and plays an important role in normal physiological functions. Iron is involved in a variety of physiological processes in cells, such as oxygen transport, mitochondrial respiration, DNA replication, and cell signaling. However, the bioavailability of iron is limited because it mainly exists in the form of Fe3+ ions and is insoluble in aqueous solution.
Excess free Fe2+ ions may trigger oxidative stress and lipid peroxidation. The amount of iron in and out of cells is controlled by a regulatory system, with transferrin import and iron transporter export. Increasing the iron content of the variable iron pool increases susceptibility to oxidative damage and ferroptosis. Through the inhibition or induction of ferroptosis, ferroptosis may be a potential target for diagnosis, prevention and treatment in liver diseases such as liver fibrosis.
Ferroptosis and Liver Fibrosis
Fibrotic diseases develop from complex pathological processes in which cells undergo continuous. Chronic damage, and ferroptosis-related mechanisms play an important regulatory role in the progression of fibrotic diseases. The main pathological features of liver fibrosis are the activation of HSCs and excessive deposition of ECM in the liver, and the abnormal proliferation of a large number of fibrous connective tissues in the portal area, thereby destroying the normal structure and physiological functions of the liver.
Therefore, inhibiting the activation and proliferation of HSCs and inducing the death of HSCs is an effective method for the treatment of liver fibrosis. Studies have shown that HSCs store a large amount of iron ions. Ferroptosis can affect the development of liver fibrosis by regulating the iron ion content in HSCs and the degree of lipid peroxidation. Therefore, targeting HSC ferroptosis may become a new strategy for the treatment of liver fibrosis.
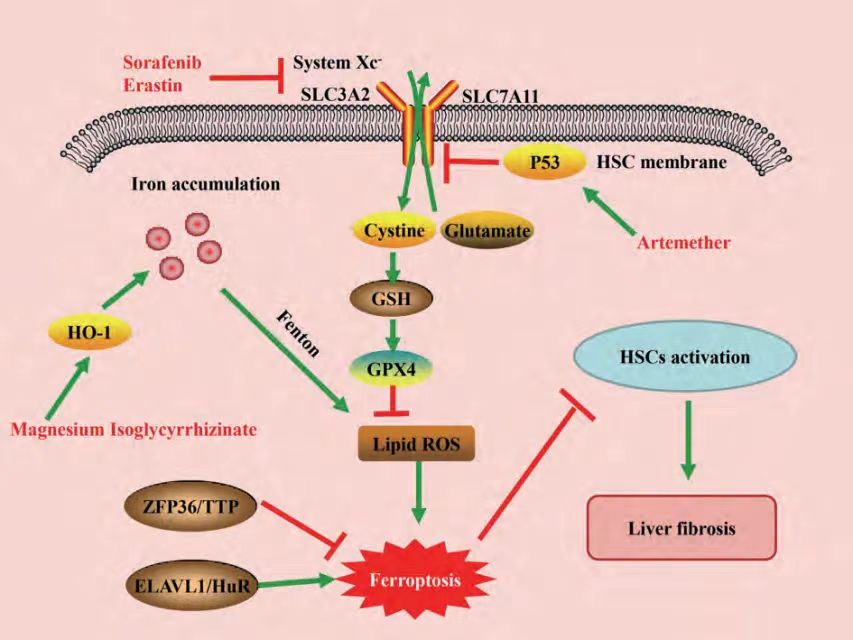
Figure 1: Regulatory pathways related to ferroptosis in liver fibrosis
The role and molecular mechanism of ferroptosis in liver fibrosis
1 RNA-binding proteins ZFP36/TTP and ELAVL1/HuR regulate ferroptosis
Sui etc[2]found that in a rat liver fibrosis model, magnesium isoglycyrrhizinate can induce the upregulation of HO-1, promote the accumulation of iron and lipid peroxides, thereby promoting the occurrence of ferroptosis in HSC; while the anti-fibrosis effect of magnesium isoglycyrrhizinate can be eliminated by the ferrostatin-1 inhibitor or silencing HO-1.
Zinc finger protein 36 (ZFP36 ring finger protein 36, ZFP36/TTP) and embryonic lethal abnormal vision-like protein 1 (ELAV like RNA binding protein 1, ELAVL1/HuR) play an important role in the regulation of HSC ferroptosis, down-regulation of ZFP36 and up-regulation of ELAVL1 can promote sorafenib/erastin-mediated HSC ferroptosis, and further reduce liver fibrosis in mice Mechanistic studies have found that ZFP36/ELAVL1 may bind to downstream target genes and affect their mRNA stability, thereby regulating the autophagy of ferritin in HSCs, releasing iron ions, and then generating a large amount of ROS through the Fenton reaction to promote ferroptosis in HSCs[3,4].
2 The BRD7-P53-SLC25A28 axis and Trf regulate ferroptosis
Zhang etc[5]found that CRISPR/cas9-mediated knockdown of BRD7 could reduce ferroptosis in HSCs, while overexpression of BRD7 mediated by specific BRD7 plasmids could promote ferroptosis in HSCs. Further studies found that increased expression of BRD7 could promote mitochondrial translocation of P53 by directly binding to the N-terminal transactivation domain of P53, thereby interacting with downstream SLC25A28 to form a complex to mediate ferroptosis.
The researchers found that specific knockout of Trf in mouse hepatocytes showed hepatic non-transferrin-bound iron accumulation, aggravating liver fibrosis mediated by high iron diet, while specific knockout of Trf and solute carrier family 39 member 14 (solute carrier family 39 member 14, SLC39A14) in mouse hepatocytes at the same time could significantly reduce liver iron accumulation and improve liver fibrosis mediated by high iron diet or carbon tetrachloride injection. Functionally, it provides a potential therapeutic target for the prevention of iron overload-induced liver fibrosis[6].
3 P62-Keap1-Nrf signaling pathway regulates ferroptosis
Current studies believe that the final execution mechanism of ferroptosis is the plasma membrane damage caused by excessive lipid peroxide accumulation, resulting in the occurrence of cell ferroptosis. Sun etc[7]reported that silencing P62 expression would increase ferroptosis induced by erastin/sorafenib in hepatocellular carcinoma (hepatocellular carcinoma, HCC) cells.
In addition, in HCC cell xenograft mouse models and cell experiments, it was found that nuclear factor E2-related factor 2 (nuclear factor erythroid 2-related factor 2, Nrf2) knockdown also showed enhanced anti-HCC effect of erastin/sorafenib. These results suggest that the P62-Kelch-like ECH-associated protein 1 (Keap1)-Nrf2 signaling pathway may play an important role in ferroptosis. Therefore, Nrf2 may be an important target involved in ferroptosis and future treatment of various liver diseases.
Ferroptosis Inducers, Inhibitors and Liver Fibrosis
1 Targeting system Xc-inducing ferroptosis
At present, research and development have found that ferroptosis inducers mainly induce ferroptosis by targeting system Xc-, GSH, GPX4, iron ions and ROS. Among them, erastin is the earliest identified specific ferroptosis inducer, which can target and inhibit the activity of system Xc-, affect the synthesis of GSH, and promote ferroptosis in various cells[8].
2 Targeting iron ions and ROS to induce ferroptosis
Studies have shown that various traditional Chinese medicines such as artemisinin and artemisinin can induce ferroptosis by increasing ROS content, thereby affecting iron metabolism, exerting anti-liver cancer effects and low toxicity, and are very promising drugs[9]. FINO2 is a kind of plakinic acid D derivatives with 1,2-dioxolane structure, which can induce ferroptosis through Fenton-like reaction, indirect inactivation of GPX4 and direct oxidation of PUFA, and its effect is stronger in tumor cells with high iron levels[10].
3 Target GSH to induce ferroptosis
Butylcysteine sulfinimide inhibits GSH synthesis rate-limiting enzyme-glutamylcysteine ligase (gluta‐mate-cysteine ligase, GCL), reduces GSH level and GPX4 activity to promote lipid peroxidation, and then induces ferroptosis in a variety of cancer cells. Cisplatin can directly combine with GSH to form a Pt (platinum)-GSH complex, leading to the inactivation of GSH and GXP4, thereby promoting ferroptosis. The researchers further used cisplatin and erastin in combination with human lung cancer cells and human colon cancer cells and found that the combined drug showed a significant synergistic anti-tumor effect[11].
4 Target lipid peroxidation to inhibit ferroptosis
Brown rice extract can improve lipid peroxidation and cytotoxicity caused by GPX4 inactivation by inhibiting ferroptosis[12]; baicalin can also inhibit ferroptosis by inhibiting lipid peroxidation and reducing free iron accumulation, and its inhibitory effect is significantly better than some typical ferroptosis inhibitors[13].
5 Target ACSL4 to inhibit ferroptosis
Thiazolidinediones, such as rosiglitazone, pioglitazone, and troglitazone, can specifically inhibit ACSL4 expression, thereby protecting cells from RSL3-induced ferroptosis and lipid peroxidation. Among them, although troglitazone has a low inhibitory effect on ACSL4, it may have inherent antioxidant activity due to its 6-chromanol structure, and it has the strongest ferroptosis inhibitory effect among thiazolidinediones[14]. Some researchers also combine nanotechnology with ferroptosis research, using nanomaterials to inhibit or induce ferroptosis. Therefore, exploring the mechanism of action of ferroptosis inducers and inhibitors and developing new drugs targeting ferroptosis are of great significance for the treatment of liver fibrosis.
ELK Biotechnology products related to ferroptosis
As a protein antibody supplier in the field of life science and medicine, ELK biotechnology can provide high-quality reagents for ferroptosis-related research. Here are some ferroptosis products listed for your reference. See the table below for details:
| Description | Cat.NO | Application | Reactivity |
| Rat TRF(Transferrin) ELISA Kit | ELK1206 | ELISA | Rat |
| Human TRF(Transferrin) ELISA Kit | ELK1300 | ELISA | Human |
| Mouse TRF(Transferrin) ELISA Kit | ELK1330 | ELISA | Mouse |
| Dog TRF(Transferrin) ELISA Kit | ELK1356 | ELISA | Dog |
| Pig TRF(Transferrin) ELISA Kit | ELK8013 | ELISA | Pig |
| Transferrin Mouse mAb | EM1231 | WB, IHC, ELISA | Human |
| Transferrin Rabbit pAb | EA188 | WB, IHC | Human |
| Transferrin rabbit pAb | ES3967 | WB;IHC;IF;ELISA | Human;Mouse;Rat |
| Human FE(Ferritin) ELISA Kit | ELK1214 | ELISA | Human |
| Rat FE(Ferritin) ELISA Kit | ELK3783 | ELISA | Rat |
| Mouse FE(Ferritin) ELISA Kit | ELK5240 | ELISA | Mouse |
| Pig FE(Ferritin) ELISA Kit | ELK6036 | ELISA | Pig |
| Dog FE(Ferritin) ELISA Kit | ELK9108 | ELISA | Dog |
| Fer rabbit pAb | ES2337 | WB;IHC;IF;ELISA | Human;Mouse;Rat |
| Ferritin heavy chain rabbit pAb | ES5400 | WB;ELISA | Human;Mouse;Rat |
| Rat GPX4(Glutathione Peroxidase 4) ELISA Kit | ELK0719 | ELISA | Rat |
| Human GPX4(Glutathione Peroxidase 4) ELISA Kit | ELK4775 | ELISA | Human |
| Mouse GPX4(Glutathione Peroxidase 4) ELISA Kit | ELK8786 | ELISA | Mouse |
| Human GRP78(Glucose Regulated Protein 78) ELISA Kit | ELK2939 | ELISA | Human |
| Mouse GRP78(Glucose Regulated Protein 78) ELISA Kit | ELK6448 | ELISA | Mouse |
| Rat GRP78(Glucose Regulated Protein 78) ELISA Kit | ELK6834 | ELISA | Rat |
| Mouse COL4(Collagen Type IV) ELISA Kit | ELK1665 | ELISA | Mouse |
| Rat COL4(Collagen Type IV) ELISA Kit | ELK1712 | ELISA | Rat |
| Human COL4(Collagen Type IV) ELISA Kit | ELK2331 | ELISA | Human |
【references】
[1] Anderson ER, Shah YM. Iron homeostasis in the liver [J]. Compr Physiol, 2013, 3: 315-330.
[2] Sui M, Jiang X, Chen J, et al. Magnesium isoglycyrrhizinate ameliorates liver fibrosis and hepatic stellate cell activation by regulating ferroptosis signaling pathway [J]. Biomed Pharmaco‐ ther, 2018, 106: 125-133.
[3] Zhang Z, Yao Z, Wang L, et al. Activation of ferritinophagy is required for the
RNA-binding protein ELAVL1/HuR to regulate ferroptosis in hepatic stellate cells [J]. Autophagy, 2018, 14: 2083-2103.
[4] Zhang Z, Guo M, Li Y, et al. RNA-binding protein ZFP36/TTP protects against ferroptosis by regulating autophagy signaling pathway in hepatic stellate cells [J]. Autophagy, 2020, 16: 1482- 1505.
[5] Zhang Z, Guo M, Shen M, et al. The BRD7-P53-SLC25A28 axis regulates ferroptosis in hepatic stellate cells [J]. Redox Biol, 2020, 36: 101619.
[6] Yu Y, Jiang L, Wang H, et al. Hepatic transferrin plays a role in systemic iron homeostasis and liver ferroptosis [J]. Blood, 2020, 136: 726-739.
[7] Sun X, Ou Z, Chen R, et al. Activation of the p62-Keap1-NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells [J]. Hepatology, 2016, 63: 173-184.
[8] Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death [J]. Cell, 2012, 149: 1060-1072.
[9] Ooko E, Saeed ME, Kadioglu O, et al. Artemisinin derivatives induce iron-dependent cell death (ferroptosis) in tumor cells [J]. Phytomedicine, 2015, 22: 1045-1054.
[10] Abrams RP, Carroll WL, Woerpel KA. Five-membered ring peroxide selectively initiatesferroptosis in cancer cells [J]. ACS Chem Biol, 2016, 11: 1305-1312.
[11] Guo J, Xu B, Han Q, et al. Ferroptosis: a novel anti tumor action for cisplatin [J]. Cancer Res Treat, 2018, 50: 445-460.
[12] Xu WH, Li CH, Jiang TL. Ferroptosis pathway andits interven‐ tion regulated by Chinese materia medica [J]. China J Chin Mater Med (中国中药杂志), 2018, 43: 4019-4026.
[13] Xie Y, Song X, Sun X, et al. Identification of baicalein as a ferroptosis inhibitor by natural product library screening [J]. Biochen Biophys Res Commun, 2016, 473: 775-780.
[14] Doll S, Proneth B, Tyurina YY, et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition [J]. Nat Chem Biol, 2017, 13: 91-98.
[15] Chen Hao, Li Xiaofeng, Wang Hua. Research progress on the mechanism of iron death in regulating liver fibrosis [J]. Acta Pharmacologica Sinica, 2019,56(11):2916-2922.DOI:10.16438/J.0513-4870.2021-0959.


