Briefly describe the regulation mechanism of ferroptosis and iron metabolism in chronic diseases

1.Iron is an essential trace element for the human body, which not only participates in the synthesis of heme, but also plays an important role in various physiological metabolisms as a prosthetic group of some key enzymes. Iron deficiency can cause iron deficiency anemia. Human hereditary hemochromatosis is a kind of iron overload disease caused by gene mutation, which leads to iron accumulation in multiple organs, skin pigmentation and lesions of multiple organs, such as type 2 diabetes mellitus, liver fibrosis, etc. More than 85% of patients with hemochromatosis carry hereditary hemochromatosis protein (HFE) mutation gene, and a small number of patients carry hepcidin (HAMP), hemojuvelin (HJV), iron efflux protein ( ferroportin 1, FPN) or transferrin receptor (transferrin receptor, TFR) 2 mutant gene, recently reported that fibroblast growth factor 6 gene mutation causes hemochromatosis and affects iron homeostasis metabolism. Iron overload can increase the risk of cancer, diabetes, neurodegenerative diseases and cardiovascular diseases, and can also cause a new iron-dependent cell death mode - ferroptosis. In 2017 and 2019, the author team of this article (Professor Wang Fuqi) found that iron homeostasis imbalance can induce ferroptosis-mediated liver and heart organ damage. The diseases caused by the imbalance of iron homeostasis are not only a nutritional problem, but also a medical problem. Studying the regulation mechanism of iron homeostasis metabolism is expected to provide a basis for the treatment of related diseases.
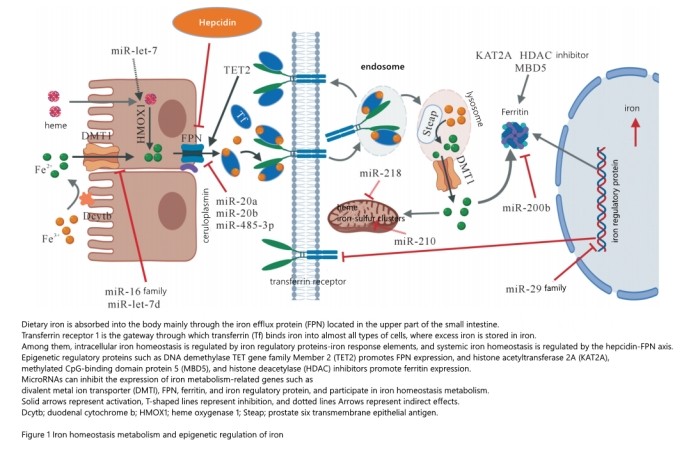
The body is mainly composed of the precise cooperation of iron absorption, utilization, storage and circulation. It strictly maintains the iron homeostasis in the system by regulating the absorption of iron in the intestine and the circulation of iron in the cells of the reticuloendothelial system. The maintenance of iron homeostasis is a complex and sophisticated system and process involving multiple links and multiple proteins (genes). Cellular iron homeostasis metabolism is mainly regulated by iron regulatory protein (iron regulatory protein, IRP) iron response elements, in which IRP can regulate the expression of iron absorption, transport and storage related proteins, such as TFR1, divalent metal ion transporter (divalent metal ion transporter) metal transport 1, DMT1) and ferritin, etc., see Figure 1. Iron metabolism in the body system is mainly regulated by HAMP-FPN, among which HAMP is a key regulator of iron uptake and release in tissues and is the control center of systemic iron homeostasis. During iron overload, HAMP binds to FPN on target cell membranes and internalizes and degrades FPN in lysosomes, thereby inhibiting iron uptake by enterocytes and release of iron into serum by macrophages or hepatocytes. absorption and distribution. At the molecular level, HAMP is regulated by the bone morphogenetic protein (BMP)/HJV/SMAD signaling pathway. HJV is a co-receptor of BMP; by enhancing the expression of BMP, it stimulates the SMAD signaling pathway and promotes the expression of HAMP. The process of BMP-stimulated HAMP expression is also regulated by HFE protein. In the high iron environment, transferrin competitively captures TFR1 bound to HFE, resulting in the separation of HFE from TFR1 and the binding of TFR2, thereby activating the SMAD signaling pathway and inducing the expression of HAMP. Membrane serine protease 2 (matriptase-2, also known as TMPRSS6) inhibits HAMP expression by cleaving HJV negative feedback. Any gene abnormality in the HAMP-regulated signal transduction pathway will affect the expression of HAMP and cause diseases related to iron metabolism disorders in the body. In recent years, with the deepening of iron metabolism mechanism research, more and more research results show that epigenetics plays an important role in iron homeostasis metabolism.
2.Iron homeostasis metabolism and histone modification regulation
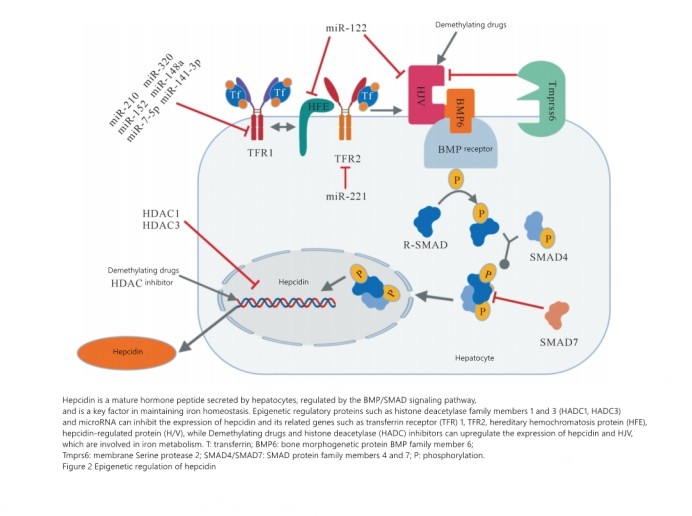
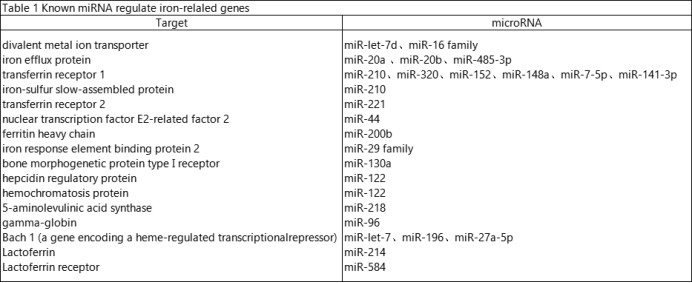
3.Iron homeostasis metabolism and microRNA regulation
4. Ferroptosis is associated with chronic disease
Chronic diseases refer to non-infectious, long-term and persistent diseases, including cardiovascular and cerebrovascular diseases, tumors, diabetes, and chronic respiratory diseases. According to WHO statistics, about 41 million people worldwide died of chronic diseases in 2016, accounting for 71% of all deaths. The latest statistics from the Chinese Center for Disease Control and Prevention show that in 2017, the number of deaths due to chronic diseases accounted for 88% of the total number of deaths. The occurrence and development of chronic diseases are not only complex in pathological mechanisms, but also affected by many factors such as heredity, environment and lifestyle. For a long time, people lacked a clear understanding of the molecular mechanism of chronic disease pathogenesis and lack of effective therapeutic drugs. In 2012, the discovery of a new type of cell death, ferroptosis, provided a new perspective for studying the occurrence, development and prevention of chronic diseases.
Ferroptosis is an iron-dependent, non-regulated form of cell death, which is different from apoptosis, pyroptosis or necrosis in terms of morphology, genetics, metabolism and molecular biology. Its main characteristics are It is the excess accumulation of lipid peroxides and reactive oxygen species. Therefore, iron metabolism and lipid peroxidation play an important regulatory role in the ferroptosis pathway, and the possible molecular mechanisms are shown in the figure below.
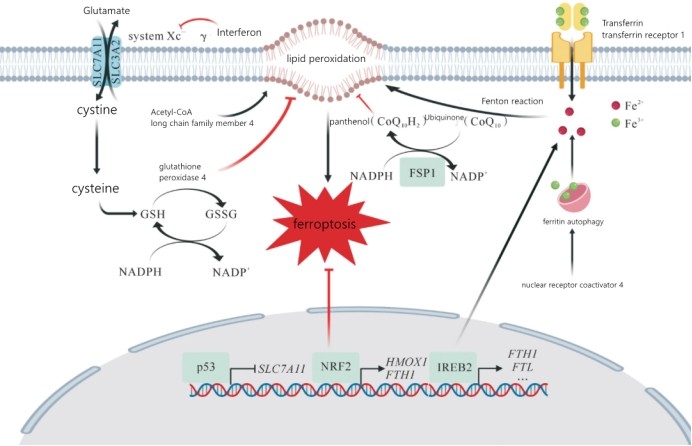
Iron metabolism and lipid metabolism pathways ultimately lead to ferroptosis by inducing lipid peroxidation, while glutathione peroxidase and ferroptosis suppressor 1 (FSP1) inhibit ferroptosis by inhibiting lipid peroxidation. FTH1: ferritin heavy chain; FTL: ferritin light chain; GSH: reduced glutathione; GSSH: oxidized glutathione; HMOX1: heme oxygenase 1; NADP+: oxidized nicotinamide adenine diamine Nucleotide phosphate; NADPH: reduced nicotinamide adenine dinucleotide phosphate; NRF2: nuclear factor E2-related factor 2; SLC7A11: solute carrier family 7 member 11; system Xc-: glutamate/cystine reverse Transporter; IREB2: Iron Response Element Binding Protein 2.
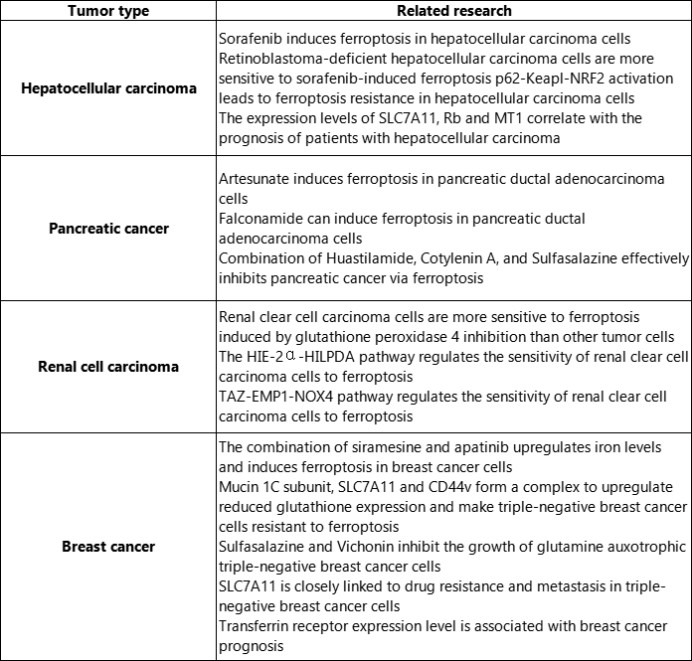
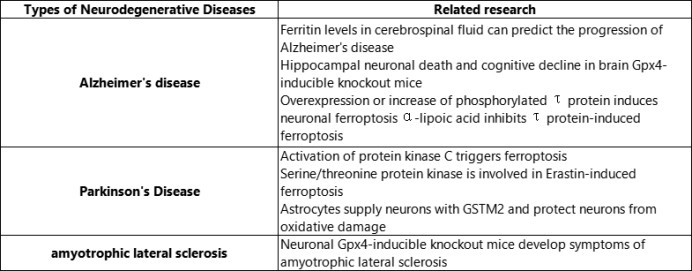
With the deepening of research, it has been found that ferroptosis plays an important role in the occurrence and development of major chronic diseases. The latest research progress of ferroptosis in tumors, neurodegenerative diseases and cardiovascular and cerebrovascular diseases.
SLC7A11: solute carrier family 7 member 11; Rb: retinoblastoma; MT1: metallothionein 1.
GPX4: glutathione peroxidase 4; GSTM2: glutathione S-transferase Mu2.
Ferroptosis and cardiovascular and cerebrovascular diseases
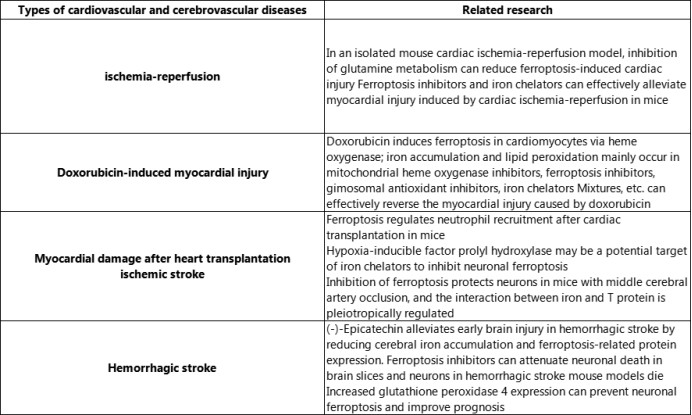
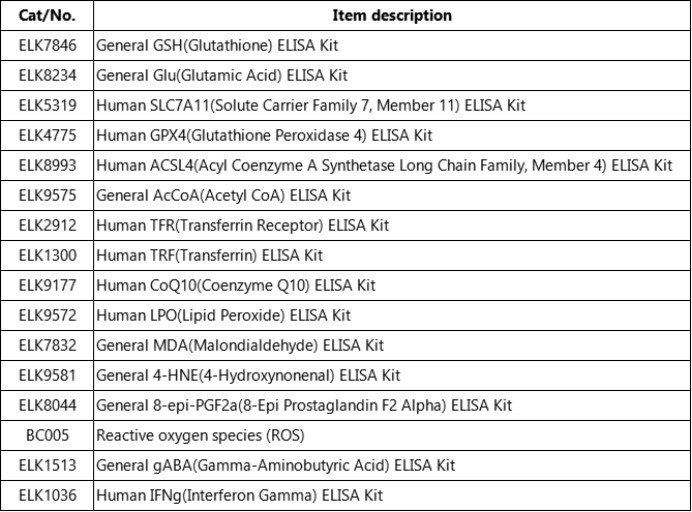
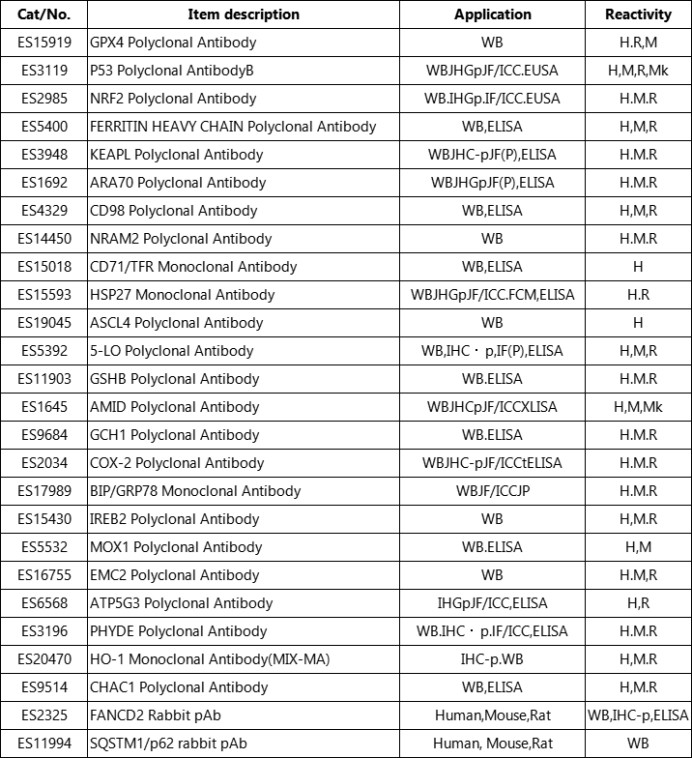
5.ELK Biotechnology provides some ferroptosis related products